Brady EMEA and Caretag Announce Strategic Partnership for End-to-End Surgical Instrument Tracking
A new partnership combines Brady EMEA’s identification technology with Caretag’s UHF RFID platform to deliver end-to-end surgical instrument management for hospitals.
www.brady.eu
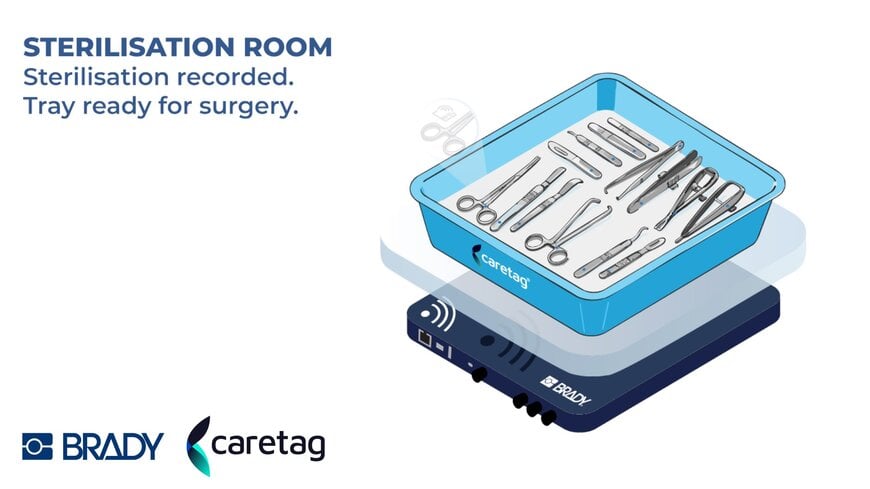
Brady EMEA, a provider of identification solutions for high-consequence environments, and Caretag, a healthcare technology company specialising in surgical instrument tracking, today announced a strategic partnership to deliver a fully integrated instrument management solution to hospitals and sterile supply departments (CSSD/RUMED) across Europe.
Together, Brady EMEA and Caretag are addressing one of the most persistent challenges in modern surgical care: ensuring that every instrument is precisely identified, reliably tracked, and fully traceable from sterilisation to the operating table and back. The combined solution is designed to meet the evolving demands of EU Medical Device Regulation (MDR 2017/745) while delivering measurable gains in efficiency, safety, and sustainability.
A Critical Challenge, a Shared Commitment
Surgical instrument management remains one of the most complex and risk-laden processes in healthcare. Manual tracking methods are time-consuming and error-prone, creating risks both in the operating theatre and in compliance with regulatory requirements. Under EU law, healthcare providers face strengthening traceability obligations through Unique Device Identification (UDI) requirements, with transition deadlines extending to 2027 and 2028.
Brady EMEA and Caretag recognised a shared mission: to put the right instrument in the right place, every time with full documentation, full confidence, and zero compromise on patient safety.
The partnership addresses the critical need for precision and reliability in healthcare by combining expertise in identification with advanced tracking technology. The resulting comprehensive solution provides hospitals with a way to strengthen patient safety and improve operational efficiency.
Integrated Technology Solution
The Brady–Caretag solution brings together two complementary areas of leadership to create a seamless, all-in-one answer to instrument management:
- Brady contributes advanced, patented data capture technology for reliable instrument identification throughout sterilisation and surgical workflows. Brady’s accurate RFID tag reading is part of a complete, full stack AIDC solution that also includes high performance labelling, printing, lasering and engraving.
- Caretag provides its AI-powered UHF RFID platform and instrument tags, engineered to withstand more than 2,500 sterilisation cycles without impacting instrument performance or usability. The Caretag system enables the automatic, simultaneous identification of multiple instruments, accelerating the instrument packing process by more than 33% compared with traditional DataMatrix scanning, while delivering real-time location tracking, full usage history and actionable inventory insights.
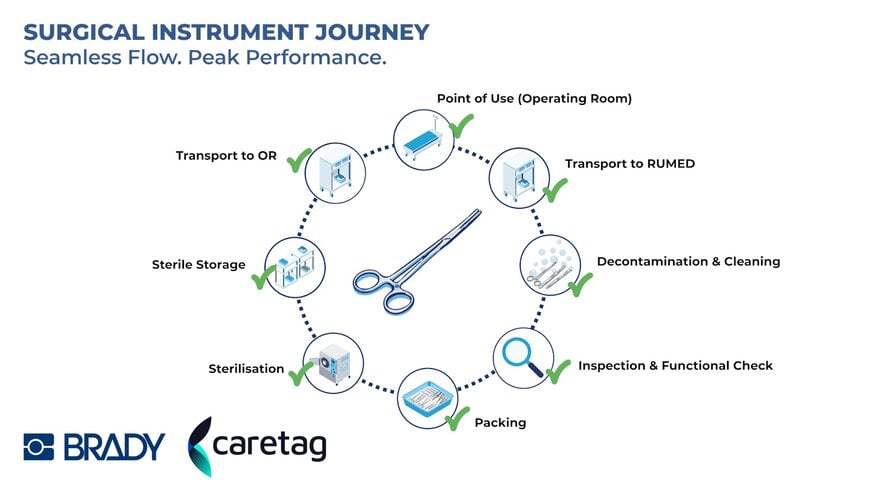
Together, the partnership enables hospitals to track every instrument throughout its complete lifecycle, from the CSSD sterile supply room to the operating theatre and back, with full digital traceability, MDR-compliant documentation, and the data insights needed to optimise instrument utilisation and reduce unnecessary procurement.
“Caretag was founded on the belief that every surgical procedure should have complete instrument traceability. Partnering with Brady EMEA allows us to extend this capability to more hospitals and healthcare systems, delivering greater transparency, efficiency and confidence in surgical instrument management”, Michael Leibig, Founder & CCO, Caretag.
Availability & Contact
The Brady EMEA–Caretag solution is available to healthcare providers across Europe. To request a demonstration, or to seek further information, please contact Michael Leibig, CCO at Caretag, at This email address is being protected from spambots. You need JavaScript enabled to view it..
Key Benefits of the Brady–Caretag Partnership
- Full instrument lifecycle traceability — from sterilisation to surgical use and return, with complete digital documentation
- Simultaneous bulk tray verification using UHF RFID, delivering a packing process more than 33% faster than DataMatrix scanning
- Real-time location tracking and usage history to prevent instrument loss and ensure surgical readiness
- Instrument utilisation data to optimise inventory, reduce waste, and support sustainability goals
- Proven durability: identification technology validated to 2,500+ sterilisation cycles with no impact on instrument performance
- Full alignment with EU MDR 2017/745 UDI requirements, with transition deadlines to 2027–2028

